COVID-19 Facts
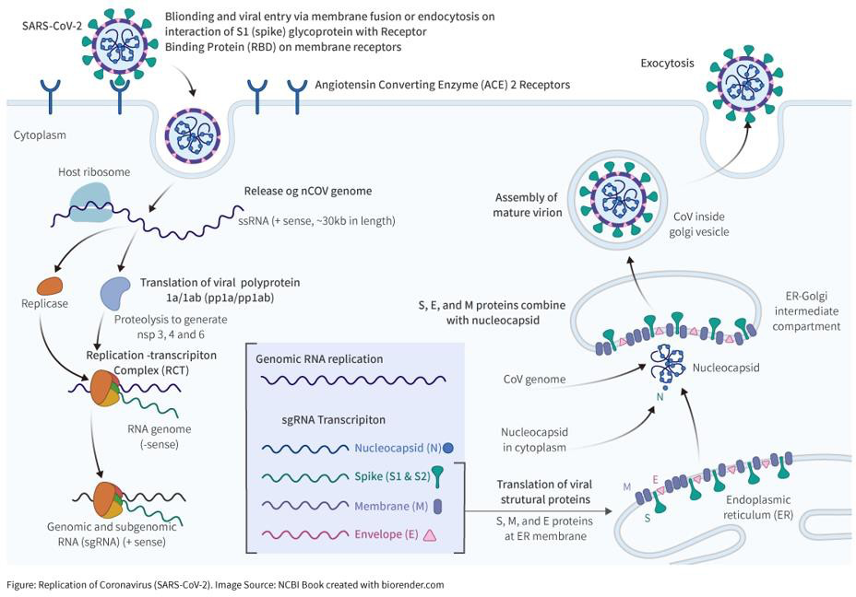
FUNDAMENTAL STRUCTURE
Club-shaped glycoprotein spikes in the envelope give the viruses a crownlike, or coronal, appearance.
The nucleocapsid, made up of a protein shell known as a capsid and containing the viral nucleic acids, is helical or tubular. The coronavirus genome consists of a single strand of positive-sense RNA [ribonucleic acid).
The N antigen (N protein) is a component protein of nucleocapsid of SARS-Cov-2, with molecular weight of about 46kDa. It is highly conserved and rich in the virus. Currently, N protein is an ideal detection marker of SARS-COV-2.
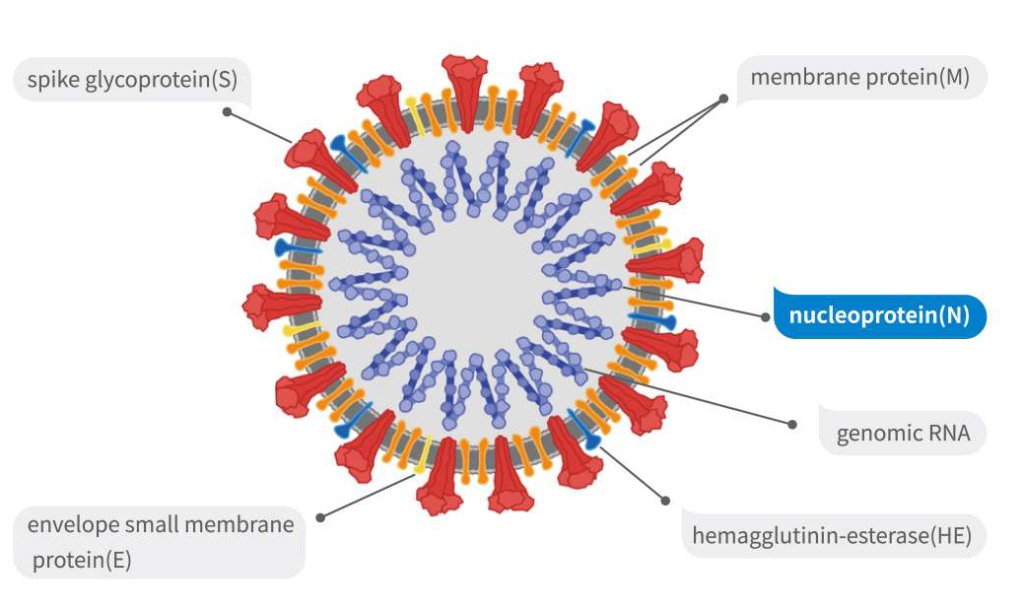
REPLICATION IN THE CELL
SYMPTOMS OF COVID-19 PATIENTS

DIAGNOSTIC METHODS
qPCR Nucleic Acid Detection
PCR nucleic acid detection can be used to diagnose COVID-19 in the early stage, which is a common diagnostic method at present. But the detection process is complex, and there will be false negative and false positive problems.
Main Reasons for False Negative:
- Sampling Deviation
- Low Viral Load
- Difference in different course and parts
Main Reason of False Positive:
The experimental process is complex and prone to cross contamination.

SARS-CoV-2 Antigen Detection